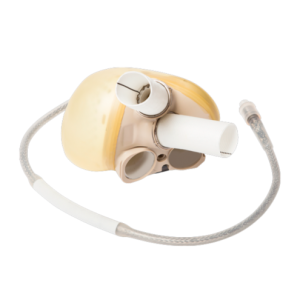
OEM & Development
With 40 years of experience in the production of cardiovascular bioprostheses and the development of consolidated products in the market, such as TAVI and other technologies, Labcor qualifies Labcor to contribute to the development of new products from their conception to prototyping and manufacturing, all of this strictly following international standards and under ISO 13485:2016 certification.
Installed in a physical area of more than 3000m² and with a classified Clean Room of more than 750m², it has an operational manufacturing capacity of more than 1500 products per month. Labcor offers the opportunity for OEM (Original Equipment Manufacturer) model services, whether or not associated with the incorporation of its own components, as well as services in regulatory, engineering, microbiological testing and validation areas.

Tissue Engineering
Labcor has the logistical capacity to capture bovine and porcine tissues, more specifically heart valves, blood vessels and pericardium, and carry out various preservation treatments either proprietary to Labcor or specified by the customer. All fabric collected by Labcor is removed from slaughterhouses certified by the Ministry of Agriculture of Brazil under federal inspection and strict environmental and operational conditions.
Our Bovine Pericardium meets the European guideline for the control and transmission of BSE (Bovine Spongiform Encephalopathy), being certified by the European Directorate for the Quality of Medicines, having the EDQM seal. Efficient collection, preparation and transport with temperature and time control and prevention of contamination are key factors for the quality of the fabric.


CLIENTES E PARCEIROS